What does a Regulatory Affairs Director do?
The Regulatory Affairs Director leads a team dedicated to ensuring that a company's products comply with all necessary regulations. This role involves working closely with government agencies, industry groups, and internal teams to navigate complex laws and guidelines. Directors must stay updated on changes in regulatory requirements and implement strategies to achieve compliance. They also play a key role in preparing and submitting documentation for regulatory approvals.
In this position, the director often liaises with other departments such as legal, quality assurance, and product development. They ensure that all products meet safety standards and that the company adheres to best practices. Directors may also be responsible for training staff on regulatory issues and coordinating responses to regulatory audits. This job requires strong analytical skills, attention to detail, and the ability to communicate effectively with various stakeholders.
How to become a Regulatory Affairs Director?
A Regulatory Affairs Director plays a key role in ensuring that products comply with government regulations. This career path requires a blend of education, experience, and specific skills. Following a clear process can help individuals achieve this important role.
Interested professionals can follow a structured path to become a Regulatory Affairs Director. The journey involves several steps, each critical to gaining the necessary knowledge and experience. Here are five essential steps:
- Earn a Relevant Degree: Start with a bachelor’s degree in a relevant field such as life sciences, health sciences, or business. A graduate degree can also be beneficial.
- Gain Experience: Work in roles related to regulatory affairs or compliance. Experience in the industry where you wish to work is highly valuable.
- Develop Key Skills: Focus on skills such as analytical thinking, attention to detail, and excellent communication. Understanding regulatory guidelines and laws is also important.
- Obtain Certifications: Consider certifications such as Certified Regulatory Affairs Professional (CRAP) to boost your credentials.
- Network and Apply: Connect with professionals in the field. Attend industry conferences and join professional associations. Apply for positions that match your skills and experience.
How long does it take to become a Regulatory Affairs Director?
Regulatory Affairs Directors play a key role in the healthcare and pharmaceutical industries. They ensure that companies follow laws and regulations. This job requires a mix of education, experience, and skills. Most often, it takes several years to reach this position.
A bachelor’s degree in life sciences, health administration, or a related field is usually the starting point. Some companies may prefer a master’s degree. Gaining experience in regulatory affairs is crucial. Many start as regulatory affairs specialists. Positions like this help build the necessary skills and knowledge. Regulatory Affairs Directors must be familiar with laws and industry standards. They must also have strong communication skills. Typically, this career path requires 5-10 years of experience. This includes working in roles that focus on compliance and regulatory matters.
Regulatory Affairs Director Job Description Sample
We are seeking a seasoned Regulatory Affairs Director to lead our regulatory strategy and compliance efforts. The ideal candidate will possess extensive experience in regulatory affairs within the industry, ensuring our products and operations align with all relevant laws and regulations.
Responsibilities:
- Develop and implement regulatory strategies to ensure compliance with local, national, and international regulations.
- Oversee the preparation and submission of regulatory filings and documentation.
- Collaborate with cross-functional teams to ensure regulatory requirements are integrated into product development and operations.
- Stay abreast of regulatory changes and assess their impact on the organization.
- Maintain relationships with regulatory agencies and act as the primary point of contact for inspections and audits.
Qualifications
- Bachelor’s degree in Life Sciences, Pharmacy, or a related field; advanced degree preferred.
- Minimum of 10 years of experience in regulatory affairs, with a minimum of 5 years in a leadership role.
- Proven track record in managing regulatory submissions and interactions with regulatory agencies.
- Strong understanding of regulatory requirements relevant to the industry.
- Excellent communication, leadership, and interpersonal skills.
Is becoming a Regulatory Affairs Director a good career path?
A Regulatory Affairs Director oversees the compliance of products and services with industry regulations. This role involves ensuring that a company adheres to all legal requirements set by governmental and industry bodies. A Regulatory Affairs Director must stay informed about laws and regulations, often requiring a background in science, engineering, or law. The position demands strong analytical skills and the ability to communicate effectively with regulatory bodies and internal teams.
This career path offers several advantages. It plays a critical role in ensuring that products meet safety and quality standards, which can enhance consumer trust. Regulatory Affairs Directors often have the opportunity to influence policy and industry standards. They typically work in stable industries such as pharmaceuticals, medical devices, and food safety. The role can also lead to significant career advancement and higher salary potential. However, there are challenges to consider. Regulatory Affairs Directors may face high pressure and tight deadlines, especially during product launches or regulatory audits. The job requires continuous learning to keep up with changing regulations. Additionally, the role may involve a significant amount of documentation and regulatory writing.
Here are some pros and cons to consider:
- Pros:
- Influence on product safety and quality
- Stable industry opportunities
- Potential for career advancement
- Higher salary potential
- Cons:
- High pressure and deadlines
- Continuous need for learning
- Extensive documentation and writing
What is the job outlook for a Regulatory Affairs Director?
The job outlook for Regulatory Affairs Directors is promising for job seekers aiming to enter this field. According to the Bureau of Labor Statistics (BLS), the industry expects to see about 94,400 job positions annually. This stability indicates a steady demand for professionals in this role. Furthermore, the BLS projects a 3.3% increase in job openings from 2022 to 2032, suggesting a positive trend in employment opportunities.
Regulatory Affairs Directors play a crucial role in ensuring compliance with laws and regulations in various industries, including pharmaceuticals, medical devices, and food products. This responsibility makes them indispensable to companies that must navigate complex regulatory landscapes. With the increasing importance of regulatory compliance, more companies are seeking skilled directors to manage these aspects, further bolstering job prospects for professionals in this field.
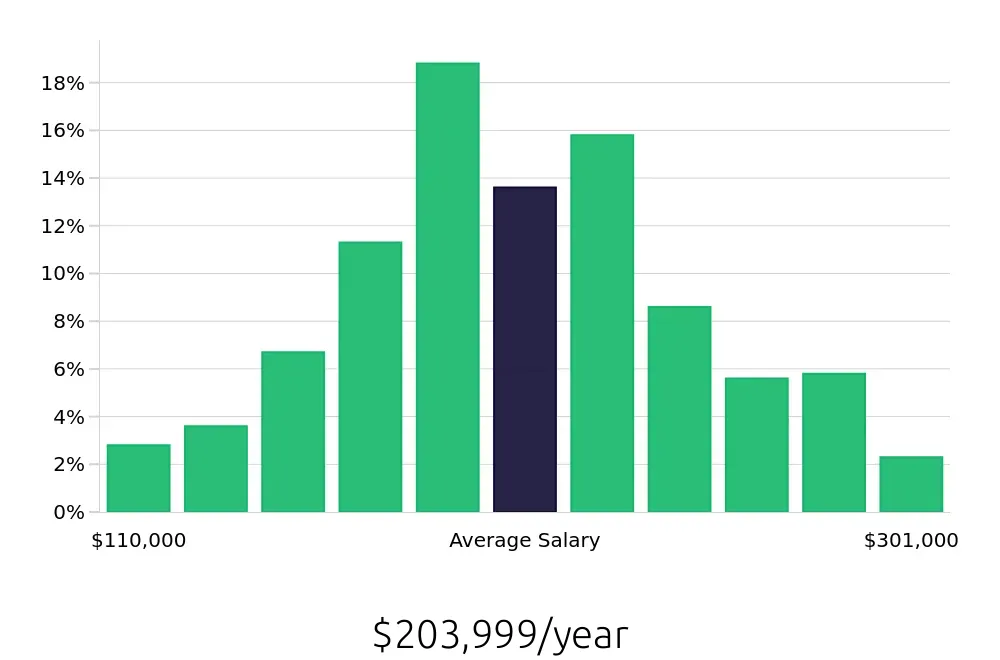
For those considering a career as a Regulatory Affairs Director, the average national annual compensation stands at $146,320, with an hourly rate of $70.35, as reported by the BLS. This compensation reflects the value and expertise required in this role. Job seekers will find that not only is the job outlook positive, but the compensation is competitive, making it an attractive career path.
Currently 141 Regulatory Affairs Director job openings, nationwide.
Continue to Salaries for Regulatory Affairs Director