What does a Regulatory Affairs Manager do?
The Regulatory Affairs Manager plays a crucial role in ensuring that a company complies with all relevant regulations and standards. This position involves working closely with government agencies and regulatory bodies to understand and implement necessary rules. It is important for the success of the organization that all products and services meet legal requirements.
This manager collaborates with different departments, including product development, marketing, and quality assurance. They develop and maintain policies and procedures that align with regulatory needs. Their expertise helps the company avoid penalties and maintain a good reputation. This role requires strong communication skills and a detailed understanding of regulatory environments.
How to become a Regulatory Affairs Manager?
Becoming a Regulatory Affairs Manager involves several steps that require dedication and the right qualifications. This role is key in ensuring that products comply with local and international regulations. Understanding the path to this position can help guide aspiring professionals. Below is an outline of the process to become a Regulatory Affairs Manager.
One must first obtain a solid educational background in a related field. This usually means earning a bachelor’s degree in life sciences, engineering, or a related discipline. Some employers may prefer or require a master's degree for more advanced positions. Gaining relevant experience is the next crucial step. This can be achieved through internships, entry-level jobs, or positions in related industries. Experience helps build a strong foundation in regulatory practices and industry standards.
To move forward, professionals should obtain relevant certifications. Certifications from organizations such as the Regulatory Affairs Professionals Society (RAPS) can boost credibility and knowledge. Networking with industry professionals and attending related conferences also enhances opportunities. This networking can lead to job leads and professional growth. Securing a position as a Regulatory Affairs Specialist or Coordinator is often the starting point. This role offers hands-on experience and the chance to understand regulatory processes more deeply.
- Earn a bachelor’s degree in a related field.
- Gain relevant experience through internships or entry-level jobs.
- Obtain certifications from recognized organizations.
- Network with industry professionals and attend conferences.
- Start in a position as a Regulatory Affairs Specialist or Coordinator.
How long does it take to become a Regulatory Affairs Manager?
The journey to becoming a Regulatory Affairs Manager involves a blend of education and experience. Most managers hold at least a bachelor's degree, often in science, business, or a related field. This degree usually takes four years to complete.
After earning a degree, gaining relevant experience is essential. Many professionals start in entry-level positions within regulatory affairs or related fields. On average, it takes about three to five years to gather the necessary experience. This period includes roles like Regulatory Affairs Specialist or Compliance Officer, where one can build skills and knowledge.
Some professionals choose to further their education with a master's degree or specialized certifications. This can add another one to two years to the timeline. Networking and professional development also play a role in advancing one's career. These steps can help in transitioning to a Regulatory Affairs Manager position.
Regulatory Affairs Manager Job Description Sample
We are seeking an experienced Regulatory Affairs Manager to join our team. The ideal candidate will have a strong background in regulatory affairs, with a focus on healthcare, pharmaceuticals, or related industries. This role involves ensuring compliance with regulatory requirements, managing product approvals, and coordinating with cross-functional teams to facilitate the successful launch of new products.
Responsibilities:
- Develop and implement regulatory strategies to ensure compliance with local and international regulatory requirements.
- Manage the regulatory approval process for new products, including preparation and submission of regulatory documentation.
- Coordinate with cross-functional teams such as clinical, quality, and marketing to ensure timely and accurate regulatory submissions.
- Stay updated on regulatory changes and trends, and provide recommendations to management.
- Prepare and deliver regulatory-related training and presentations to internal stakeholders.
Qualifications
- Bachelor's degree in a relevant field (e.g., Life Sciences, Pharmacy, Business Administration).
- Minimum of 5-7 years of experience in regulatory affairs, preferably in the healthcare or pharmaceutical industry.
- Strong knowledge of regulatory requirements and guidelines (e.g., FDA, EMA, MHLW).
- Excellent organizational and project management skills.
- Strong analytical and problem-solving skills.
Is becoming a Regulatory Affairs Manager a good career path?
A Regulatory Affairs Manager oversees the compliance of products within industries such as pharmaceuticals, food, and medical devices. This role involves ensuring that these products meet all necessary regulations and standards. It requires a keen understanding of regulatory environments and an ability to communicate effectively with regulatory bodies.
The career path for a Regulatory Affairs Manager offers several benefits. For one, it provides a strong sense of purpose, as this role directly impacts public safety. It also offers competitive salaries and the chance to work with cutting-edge technology. However, it comes with challenges such as managing strict deadlines and navigating complex regulations. Regulatory Affairs Managers must stay updated with changes in laws, which can be demanding. Balancing these pros and cons can help potential candidates decide if this path suits them.
Here are some pros and cons to consider:
- Pros:
- Job security: Regulatory Affairs Managers are essential in regulated industries.
- Competitive salaries: This role often offers good compensation.
- Opportunity for advancement: Experienced managers can move into higher positions.
- Cons:
- High stress: Meeting deadlines and ensuring compliance can be challenging.
- Complex regulations: Constant updates in laws require continuous learning.
- Limited travel: Working with regulatory bodies may limit travel opportunities.
What is the job outlook for a Regulatory Affairs Manager?
The job outlook for Regulatory Affairs Managers looks promising, with an expected annual average of 94,400 job positions available from the Bureau of Labor Statistics (BLS). This indicates a robust demand for professionals in this field. With a projected growth of 3.3% from 2022 to 2032, this role continues to be in steady demand across industries.
A Regulatory Affairs Manager plays a crucial role in ensuring compliance with laws and regulations. This position involves working with government agencies, advocating for industry standards, and managing product approvals. This responsibility makes Regulatory Affairs Managers valuable assets to companies. Their expertise helps ensure products meet necessary regulatory requirements, which is vital in a rapidly evolving regulatory landscape.
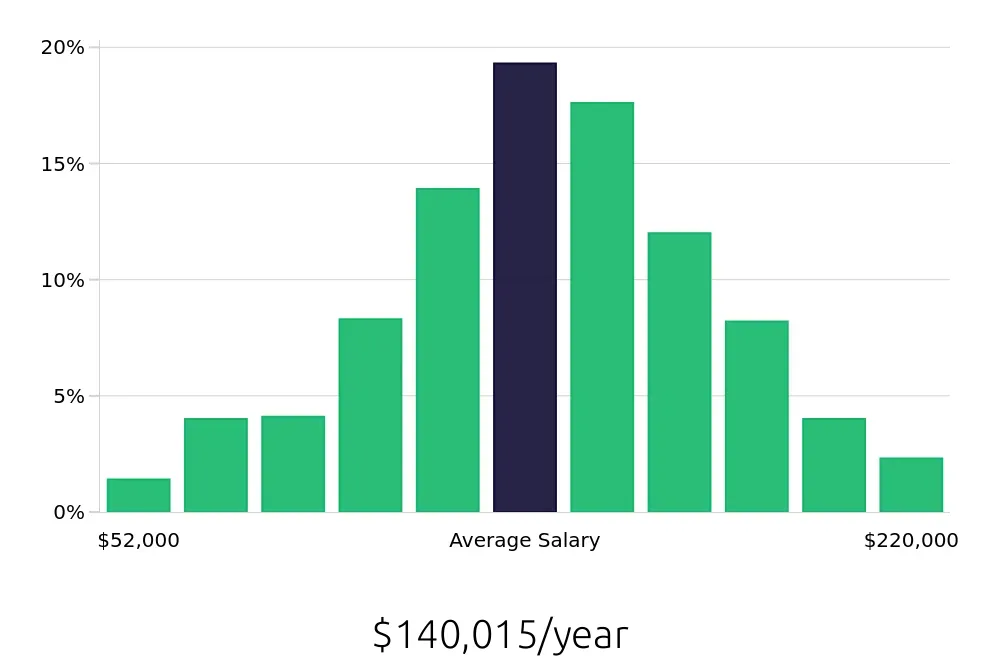
Professionals in this field also enjoy competitive compensation. The average national annual salary for a Regulatory Affairs Manager is $146,320, according to the BLS. On an hourly basis, the average compensation is $70.35. This indicates that Regulatory Affairs Managers not only contribute significantly to their companies but also receive fair financial rewards for their expertise and dedication. This makes it an attractive career choice for job seekers looking for both stability and a good salary.
Currently 133 Regulatory Affairs Manager job openings, nationwide.
Continue to Salaries for Regulatory Affairs Manager