What does a Clinical Research Manager do?
A Clinical Research Manager plays a key role in the world of medical research. They oversee clinical trials and research studies to ensure they meet strict guidelines. This role involves working with doctors, scientists, and other healthcare professionals. The manager coordinates schedules, gathers data, and ensures everything runs smoothly. They must be very organized and detail-oriented. A successful manager ensures research is conducted ethically and efficiently.
In this position, the manager must understand both medical research and project management. They prepare budgets, manage resources, and report findings to stakeholders. Communication is crucial, as they often work with a diverse team. The manager must explain complex medical information in an understandable way. Attention to detail helps in preventing errors and maintaining high standards. This role is essential for advancing medical knowledge and improving patient care.
How to become a Clinical Research Manager?
Becoming a Clinical Research Manager can open the door to an exciting career in the healthcare industry. This role involves overseeing clinical trials and ensuring they follow all guidelines. A Clinical Research Manager plays a key part in medical advancements and patient care.
Here are the steps to take towards this fulfilling career:
- Earn a bachelor’s degree in a relevant field. Many Clinical Research Managers start with degrees in science, nursing, or a related area.
- Gain experience through internships or entry-level positions. Working in clinical research helps build the skills and knowledge needed for this role.
- Consider a master’s degree. Advanced degrees can make a candidate more competitive in the job market.
- Obtain certification. Certifications such as CRA (Certified Clinical Research Associate) can boost a resume and show expertise.
- Seek managerial experience. Working in a supervisory role in research settings can provide the leadership skills needed for management.
How long does it take to become a Clinical Research Manager?
The journey to becoming a Clinical Research Manager involves several steps and a solid educational background. Typically, candidates start with a bachelor's degree in a field related to health or science, such as biology or chemistry. This foundational degree usually takes four years to complete.
After earning a bachelor's degree, many professionals gain experience through entry-level positions in clinical research. They might work as clinical research assistants or coordinators. This hands-on experience can last from one to three years, depending on the individual's career progression. Some may choose to further their education by pursuing a master's degree in clinical research or a related field. This additional degree adds to the time, usually taking another two to three years to complete. Finally, gaining leadership experience and accumulating several years in the field allows a professional to step into a Clinical Research Manager role.
Overall, the timeline to become a Clinical Research Manager can range from six to nine years. This duration includes time spent on education, gaining experience, and developing leadership skills. Each step is crucial for building the necessary expertise and confidence to manage clinical research projects effectively.
Clinical Research Manager Job Description Sample
A Clinical Research Manager is responsible for overseeing and coordinating clinical research projects from inception to completion. This role involves managing a team of researchers, ensuring compliance with regulatory standards, and maintaining high-quality research practices.
Responsibilities:
- Oversee and coordinate all aspects of clinical research projects, including study design, implementation, and reporting.
- Manage and mentor a team of clinical research professionals, providing guidance and support to ensure project goals are met.
- Develop and maintain relationships with key stakeholders, including study sponsors, investigators, and regulatory agencies.
- Ensure all clinical research activities comply with applicable laws, regulations, and ethical guidelines.
- Prepare and review research protocols, informed consent forms, and other regulatory documents.
Qualifications
- Advanced degree (e.g., MD, PhD, PharmD) in a relevant scientific or healthcare field.
- Minimum of 5-7 years of experience in clinical research, with at least 3 years in a managerial or supervisory role.
- Proven track record of successfully managing complex clinical research projects.
- Strong understanding of clinical research methodologies, regulatory requirements, and ethical standards.
- Excellent leadership and team management skills.
Is becoming a Clinical Research Manager a good career path?
A Clinical Research Manager oversees clinical trials and research studies. This role requires a deep understanding of medical research and patient care. Clinical Research Managers work in hospitals, research labs, and pharmaceutical companies. They ensure that trials run smoothly and meet regulatory standards. This job offers the chance to contribute to medical advancements and improve patient care.
Being a Clinical Research Manager has many benefits. They work in a field that makes a real difference in people's lives. Managers can advance quickly and often earn a good salary. This career provides opportunities to travel for work and collaborate with a diverse team of professionals. It also offers job stability, as medical research is always in demand.
However, this role also has challenges. Clinical Research Managers often work long hours and face high stress. They must meet tight deadlines and ensure accuracy in data. The job can be demanding, requiring constant attention to detail and the ability to manage complex projects. Here are some pros and cons to consider:
- Pros:
- Contribution to medical advancements
- Opportunity for career advancement
- Job stability in a growing field
- Chance to work with diverse professionals
- Cons:
- Long hours and high stress
- Need for constant attention to detail
- High responsibility in ensuring accuracy
- Demanding work environment
What is the job outlook for a Clinical Research Manager?
Becoming a Clinical Research Manager can open doors to a promising career path. The Bureau of Labor Statistics (BLS) reports an average of 6,500 job positions available each year. This demand shows a stable and growing field for job seekers. Clinical Research Managers play a crucial role in the healthcare sector, overseeing clinical trials and ensuring the success of research projects. Their expertise is highly valued, leading to good job security and opportunities for advancement.
The outlook for Clinical Research Managers looks positive, with a projected job opening percent change of 4.8% from 2022 to 2032, according to BLS. This growth reflects the increasing importance of clinical research in healthcare. As medical advancements continue, the need for skilled professionals to manage these projects will rise. Job seekers in this field can expect a steady stream of opportunities and potential for career growth. With the right qualifications and experience, a Clinical Research Manager can look forward to a promising and rewarding career.
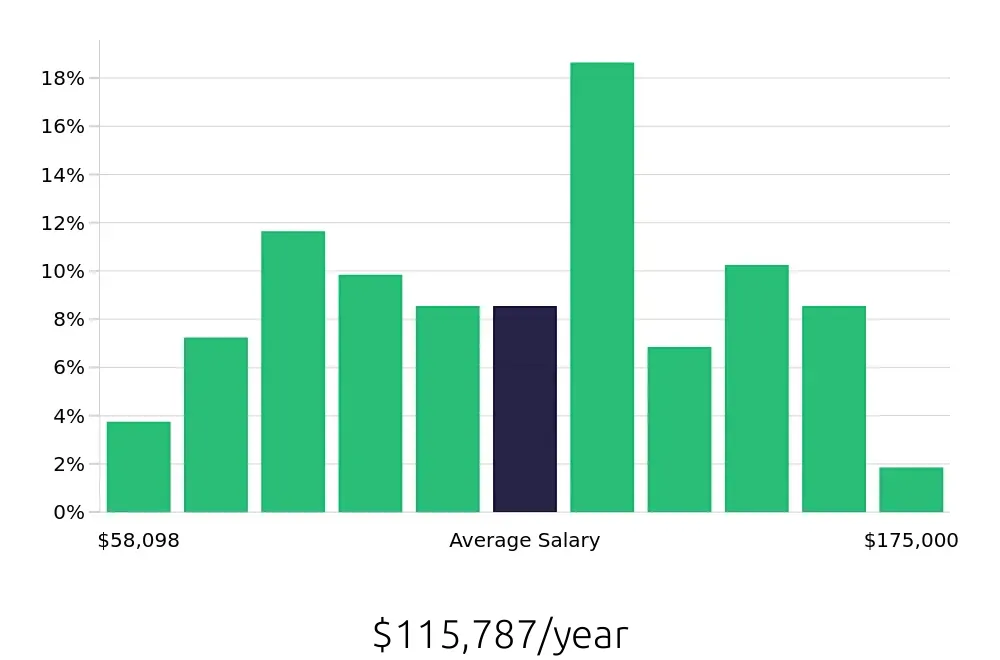
For those considering this career, the compensation is quite attractive. The average national annual salary for Clinical Research Managers stands at $169,120, per BLS data. This salary reflects the specialized skills and responsibilities that come with the role. Additionally, the hourly compensation averages at $81.31. Such figures make this a lucrative career choice for professionals in the field. Job seekers can expect competitive pay and the potential for bonuses and benefits. This financial incentive adds to the appeal of pursuing a career as a Clinical Research Manager.
Currently 133 Clinical Research Manager job openings, nationwide.
Continue to Salaries for Clinical Research Manager