What does a Regulatory Affairs Specialist do?
A Regulatory Affairs Specialist works to ensure that a company’s products meet all legal and regulatory requirements. This role involves interacting with government agencies, monitoring changes in regulations, and helping to develop strategies to comply with these laws. They gather data, prepare documentation, and often need to communicate complex information clearly. This specialist serves as a liaison between the company and regulatory bodies, facilitating smooth approvals and compliance processes.
In this position, the Regulatory Affairs Specialist manages the submission of necessary documents to ensure products are safe and effective. They may conduct research on industry standards and collaborate with other departments, such as quality assurance and product development. Keeping up-to-date with the latest regulations and ensuring all procedures are followed correctly helps to protect the company from legal issues and maintains the integrity of their products. This role demands attention to detail, strong analytical skills, and the ability to work under pressure.
How to become a Regulatory Affairs Specialist?
Becoming a Regulatory Affairs Specialist is a rewarding career path for those interested in ensuring products comply with industry regulations. This field offers a mix of science, law, and business. It requires a strong understanding of regulatory frameworks and the ability to communicate effectively with various stakeholders. By following these steps, an individual can successfully transition into this specialized role.
To embark on this career, one must first obtain a relevant educational background. This usually involves a bachelor's degree in life sciences, pharmacy, or a related field. Gaining practical experience through internships or entry-level positions in regulatory affairs can also be beneficial. Networking with professionals in the industry and attending relevant workshops or seminars can provide further insights and opportunities. Professional certifications in regulatory affairs can enhance job prospects and open doors to advanced positions. Each step in this process builds the foundation needed for a successful career as a Regulatory Affairs Specialist.
Here are five key steps to becoming a Regulatory Affairs Specialist:
- Earn a Relevant Degree: Start with a bachelor's degree in life sciences, pharmacy, or a related field. This education provides the necessary scientific knowledge.
- Gain Practical Experience: Seek internships or entry-level positions in regulatory affairs. This hands-on experience is crucial for understanding the field.
- Network with Professionals: Connect with others in the industry through professional organizations and events. Networking can lead to job opportunities and mentorship.
- Attend Workshops and Seminars: Participate in industry-specific training programs. These events can provide valuable insights and keep you updated on the latest regulations.
- Obtain Certifications: Pursue professional certifications in regulatory affairs. These credentials can enhance your resume and open doors to advanced roles.
How long does it take to become a Regulatory Affairs Specialist?
A Regulatory Affairs Specialist plays a key role in ensuring that products meet legal requirements. The time to become one varies based on education and experience. Most positions require at least a bachelor's degree. Common fields include science, business, or health administration.
The journey often involves several steps. First, a person earns a bachelor's degree, which takes about four years. Next, gaining experience through internships or entry-level jobs helps build skills. This may take one to three years. Some choose to pursue a master’s degree to advance their career, adding another one to two years. In total, it may take five to seven years to become a Regulatory Affairs Specialist. Persistence and dedication are key to success in this field.
Regulatory Affairs Specialist Job Description Sample
The Regulatory Affairs Specialist is responsible for ensuring that the company complies with all applicable regulatory requirements and standards related to products and services. This role involves working closely with various departments to manage regulatory submissions, maintain regulatory compliance, and support product development.
Responsibilities:
- Prepare and submit regulatory applications and documents to appropriate agencies.
- Maintain up-to-date knowledge of regulatory requirements and industry trends.
- Collaborate with cross-functional teams, including R&D, Quality Assurance, and Marketing.
- Conduct risk assessments and develop mitigation strategies.
- Prepare and deliver training materials and presentations on regulatory topics.
Qualifications
- Bachelor's degree in Life Sciences, Regulatory Affairs, or related field.
- Minimum of 3-5 years of experience in regulatory affairs, preferably in the pharmaceutical, biotechnology, or medical device industry.
- Strong understanding of regulatory requirements and processes, including FDA regulations.
- Excellent written and verbal communication skills.
- Ability to work independently and manage multiple tasks simultaneously.
Is becoming a Regulatory Affairs Specialist a good career path?
A Regulatory Affairs Specialist plays a critical role in ensuring that products comply with government regulations. This career path involves working with health authorities to ensure that products are safe for consumers. This role is vital in industries like pharmaceuticals, medical devices, and food. Specialists must stay updated on laws and regulations, making this a dynamic and important job.
Working in regulatory affairs offers many benefits. The job provides a sense of purpose by ensuring public safety. Specialists often work with top companies, which can lead to good job security. Networking with professionals in the industry can also open doors to new opportunities. However, the role can be demanding. Specialists must keep up with ever-changing regulations, which can be stressful. The job may also involve long hours, especially during product launches.
Here are some pros and cons to consider:
- Pros:
- High job security in regulated industries.
- Opportunities for career advancement.
- Contributing to public safety and well-being.
- Cons:
- Demanding workload with strict deadlines.
- Continuous need to stay updated on regulations.
- Potential for long working hours during critical periods.
What is the job outlook for a Regulatory Affairs Specialist?
The job outlook for Regulatory Affairs Specialists is quite positive. The Bureau of Labor Statistics (BLS) reports an average of 31,000 job positions available each year. This means there are many opportunities for qualified candidates. The BLS also projects a 4.6% increase in job openings from 2022 to 2032. This growth rate shows that companies are increasingly valuing expertise in regulatory affairs. Job seekers looking for stability and growth will find this field promising.
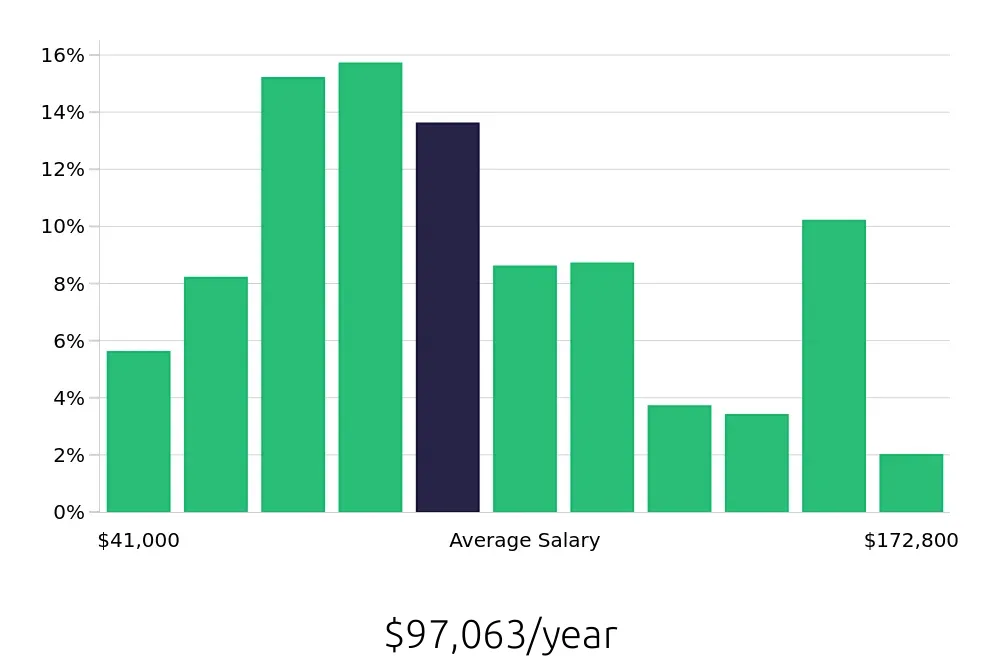
Another key aspect is the compensation. Regulatory Affairs Specialists earn a strong average national annual salary of $80,190. This figure highlights the financial rewards of this career path. Hourly compensation stands at $38.55, indicating a good rate of pay for the work involved. These figures suggest a rewarding career both in terms of job satisfaction and financial benefits. Aspiring professionals will appreciate the competitive salary and benefits package.
For those considering this career, the combination of job availability and compensation makes it an attractive option. The increasing demand for regulatory expertise across various industries ensures a steady flow of opportunities. The strong salary further adds to the appeal, making it a worthwhile career choice for many job seekers. With these factors in mind, Regulatory Affairs Specialists can look forward to a promising and rewarding future.
Currently 131 Regulatory Affairs Specialist job openings, nationwide.
Continue to Salaries for Regulatory Affairs Specialist